In 2026, the landscape of healthcare will be significantly shaped by the introduction and utilization of Api Drugs. An Api Drug, or Active Pharmaceutical Ingredient, plays a critical role in drug formulation. Recent reports from the Global Pharmaceutical Industry indicate that the market for Api Drugs is projected to reach $300 billion by 2026. This growth reflects the increasing demand for high-quality generics and biosimilars.
Dr. Emily Carter, a leading pharmaceutical expert, emphasizes, "Api Drugs are the backbone of modern medical therapies." Her insights highlight how these drugs impact patient outcomes and accessibility. Despite the advancements, challenges remain. Quality control and production scalability can often lag behind demand.
As the healthcare sector evolves, stakeholders must reflect on the balance between innovation and safety. The powerful data surrounding Api Drugs can inspire progress, yet vigilance is key. Ensuring that these ingredients meet the necessary standards will be vital for future patients.

Active Pharmaceutical Ingredients (APIs) are the core components in drug formulation. They are the active substances that provide therapeutic effects. In essence, without APIs, there would be no effective medicines. The pharmaceutical industry relies on them to develop treatments that alleviate various health conditions.
API drugs can have a significant impact on healthcare by reducing the burden of diseases. However, their production can be complex and costly. Many manufacturers face challenges in ensuring quality and consistency. There are also concerns regarding sourcing and environmental effects. Ethical practices in API production must be prioritized. Transparency in this area is crucial.
The importance of APIs cannot be understated. They hold the key to innovation in medication development. Yet, this sector sometimes lacks regulation. There are instances of subpar APIs reaching the market. This raises questions about efficacy and safety. Stakeholders must reflect on these imperfections. Enhanced collaboration and rigorous standards are necessary. This approach can improve both quality and trust in healthcare systems.
| API Drug Type | Definition | Impact on Healthcare in 2026 | Growth Rate (%) |
|---|---|---|---|
| Small Molecule APIs | Chemical compounds that constitute the active ingredient in pharmaceutical drugs. | Continued prevalence in generic drug production, lowering costs for patients. | 4.5% |
| Biologics | Large, complex molecules generally derived from living cells. | Expecting increased investment in research, leading to advanced therapies and patient treatments. | 6.2% |
| Biosimilars | Biological products highly similar to an already approved biological product. | They provide more affordable options for patients, improving access to treatment. | 8.4% |
| Peptides | Short chains of amino acids used as drugs for various conditions. | Growing use in chronic disease management, particularly diabetes, enhancing patient care. | 5.7% |
| Nucleotide and Nucleic Acid APIs | APIs that are fundamental to genetic therapies and advanced vaccines. | Key role in gene therapies, promising breakthroughs in treating genetic disorders. | 10.1% |
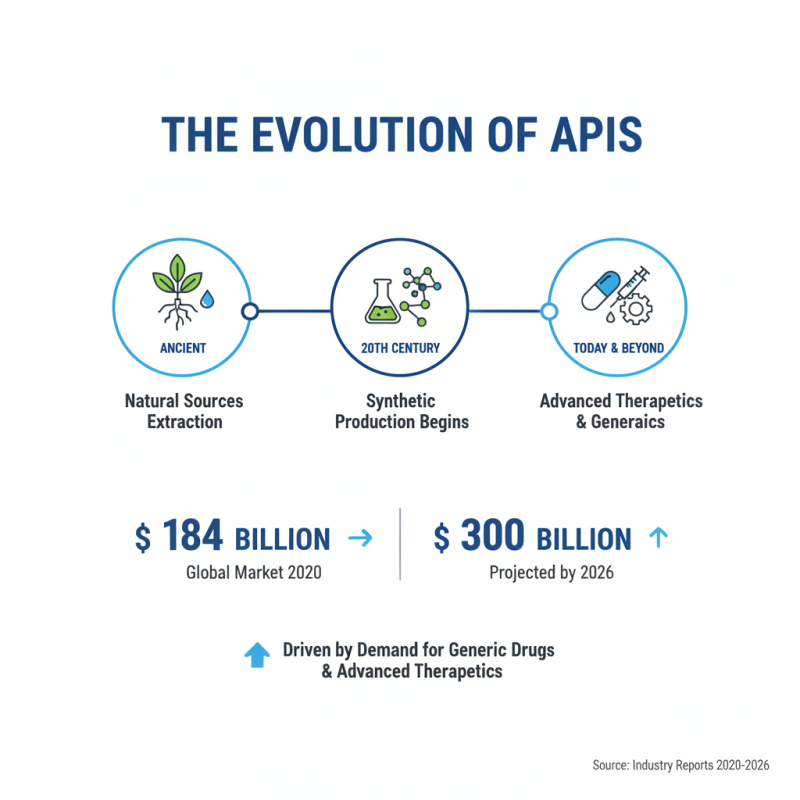
The evolution of Active Pharmaceutical Ingredients (API) has significantly shaped healthcare over the past few decades. Initially, APIs were extracted from natural sources. In the 20th century, the shift to synthetic production began, enhancing efficiency and scalability. The global API market was valued at approximately $184 billion in 2020, with projections expecting it to reach nearly $300 billion by 2026. This growth reflects the increasing demand for generic drugs and advanced therapeutics.
Key milestones in API development include the introduction of compound synthesis techniques and advanced purification processes. These innovations have made it feasible to produce complex molecules that were previously impossible. For instance, high-performance liquid chromatography (HPLC), developed in the late 1970s, remains a critical tool for ensuring purity and composition. Despite these advancements, challenges persist. Quality control remains a significant concern, often leading to recalls and regulatory scrutiny.
Moreover, the rise of biotechnology has led to the production of biologics, a new class of APIs derived from living organisms. These drugs promise tailored treatments for diseases like cancer and autoimmune disorders. However, their complexity can complicate production. The industry must balance innovation with stringent regulations to ensure patient safety. As we progress toward 2026, continuous improvement in API manufacturing practices will be crucial for advancing global healthcare.
In 2026, active pharmaceutical ingredient (API) drugs will play a crucial role in healthcare. These drugs are essential for treating various medical conditions. They directly influence patient outcomes and treatment efficacy. With advancements in drug manufacturing, the quality of APIs has improved significantly. This leads to better compatibility with patient needs.
Tips: Always consult a healthcare professional before starting a new treatment. Patient-specific factors can greatly affect outcomes.
The rise of personalized medicine is boosting the importance of APIs. Tailored treatments can result in higher efficacy rates. For instance, a specific API may work wonders for some patients but not for others. As we lean more into precision medicine, the need for high-quality APIs will grow. However, disparities in access to these drugs might raise concerns.
Tips: Keep track of your medications. Understanding your treatment plan can enhance communication with your healthcare provider.
In conclusion, while the promise of API drugs is bright, challenges remain. The landscape of healthcare is evolving, but disparities in availability should be addressed. Monitoring patient experiences and outcomes will be essential to refine these treatments further.
The regulatory landscape for Active Pharmaceutical Ingredients (API) is evolving rapidly. In 2026, compliance and safety standards are crucial for ensuring patient safety. Reports indicate that as much as 30% of drug recalls are due to API-related issues. This highlights the importance of stringent regulations. Manufacturers must adapt to these standards to avoid significant financial losses.
Implementing robust quality control measures is essential. The FDA enforces guidelines to ensure APIs are safe and effective. Yet, not all manufacturers comply fully. A survey revealed that nearly 20% of global manufacturers lack proper quality control certifications. This raises concerns over potential health risks.
Tips:
Improving API safety standards is a collective effort. Industry players must collaborate. Transparency in manufacturing processes can also build trust. If not addressed, the gap in compliance could have dire consequences for public health.

The landscape of Active Pharmaceutical Ingredients (API) is evolving rapidly. By 2026, innovations in API drug development may lead to significant shifts in healthcare. Research shows a projected compound annual growth rate (CAGR) of around 6.3% in the global API market from 2021 to 2026. This growth reflects the increasing demand for generic drugs and biologics.
One key trend is the rise of biopharmaceuticals. These drugs often require complex manufacturing processes. They can lead to higher production costs, presenting challenges for small manufacturers. Despite this, the potential benefits for patients are substantial. Advanced therapies using APIs could offer personalized treatments for chronic diseases. However, this area remains under-researched, and concerns about accessibility persist.
Sustainability is another area of focus. Many API manufacturers are exploring greener production methods. According to recent reports, around 25% of companies are prioritizing eco-friendly practices in their operations. Yet, the adoption of sustainable practices may slow due to high initial costs and regulatory complexities. Balancing innovation with accessibility and sustainability will be crucial in shaping the future of API drug development.